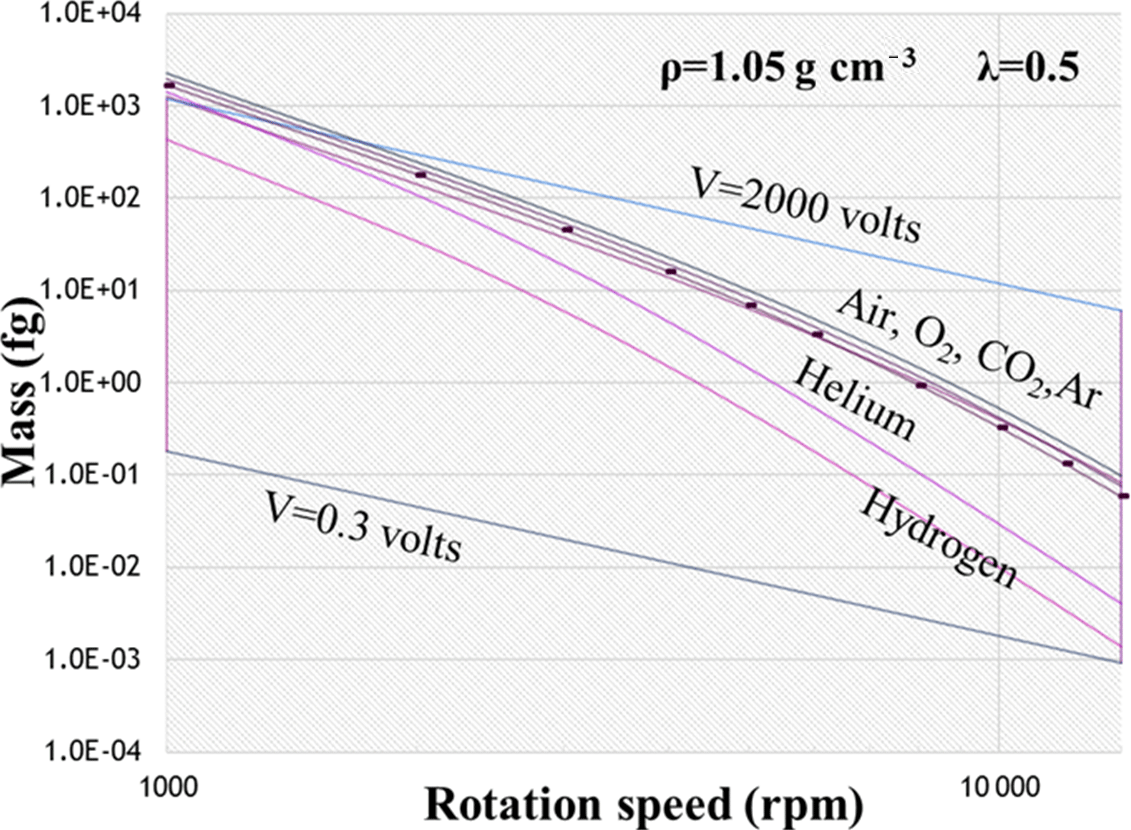
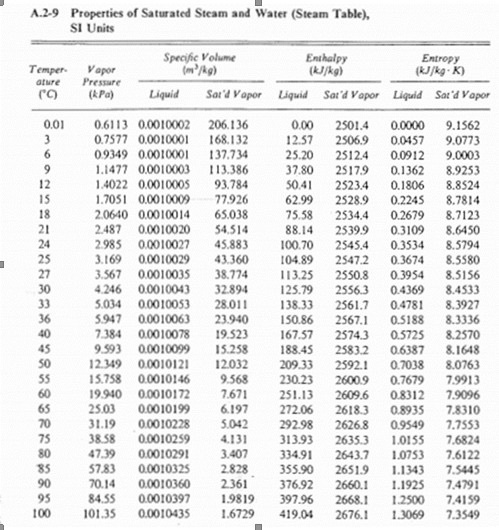
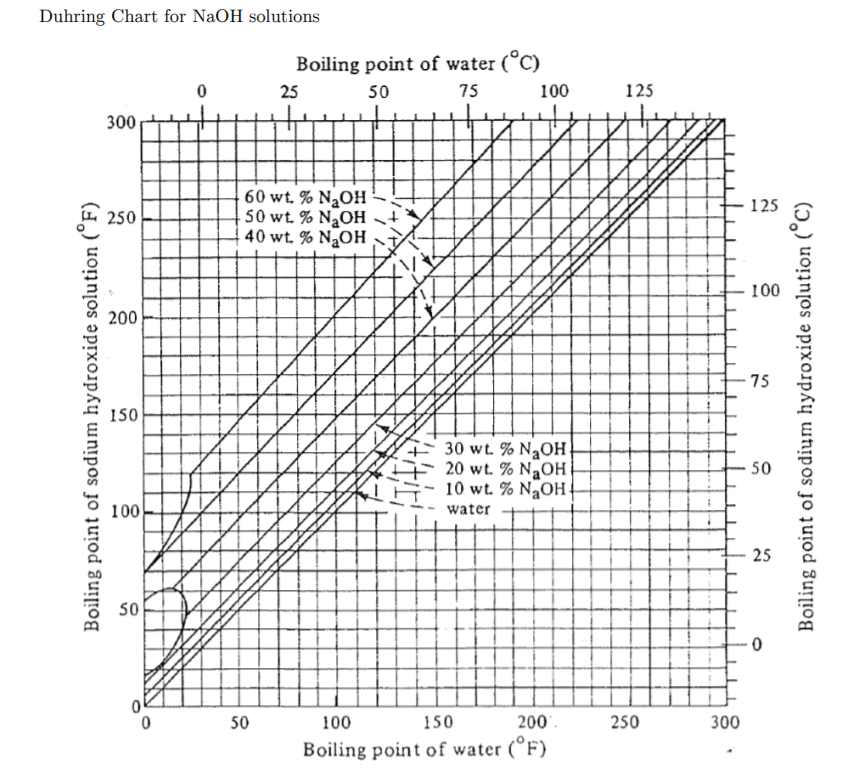
UC-600 Ammonia in Simulated Hanford Double-Shell Tank Wastes: Solubility and Effects on Surface Tension

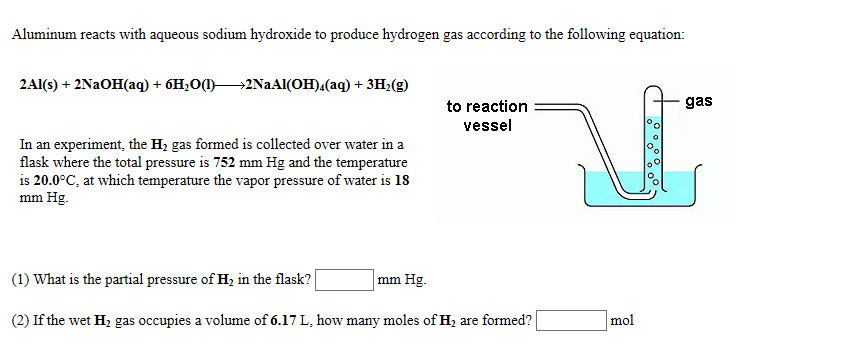
SOLVED: Sodium hydride reacts with excess water to produce aqueous sodium hydroxide and hydrogen gas. How many grams of NaH will produce 0.982 L of gas at 301 K and barometric pressure

Calculated vapor pressures of K and Na vs. temperature. For K, both... | Download Scientific Diagram
I P-V-T RELATIONS OF AQUEOUS SODIUM HYDROXIDE AND ITS CAUSTICITY AT HIGH TEMPERATURES AND HIGH PRESSURES BY Rv0 I\IYA]IA AVD SHI

SOLVED: Problem 2 (30/100): A single-effect evaporator is designed to concentrate 9,070 kg/h of a 20% solution of sodium hydroxide to 50% (mass fraction). The gauge pressure of the steam is 1.37
Solubility of Lithium Hydroxide in Water and Vapor Pressure of Solutions above 220° F. | Journal of Chemical & Engineering Data
Title P-V-T relations of aqueous sodium hydroxide and its causticity at high temperatures and high pressures Author(s) Kiyama, R

Optimization of sodium hydroxide for securing high thermoelectric performance in polycrystalline Sn1 − xSe via anisotropy and vacancy synergy - Shi - 2020 - InfoMat - Wiley Online Library
SOLUBILITY OF LITHIUM HYDROXIDE IN WATER AND VAPOR PRESSURE OF SOLUTIONS OF LITHIUM HYDROXIDE ABOVE 220 F - UNT Digital Library