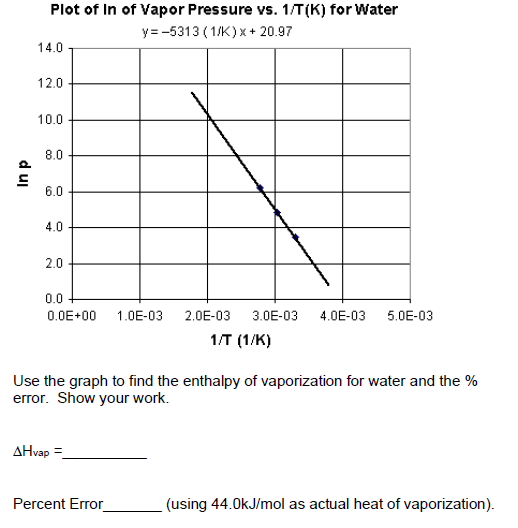
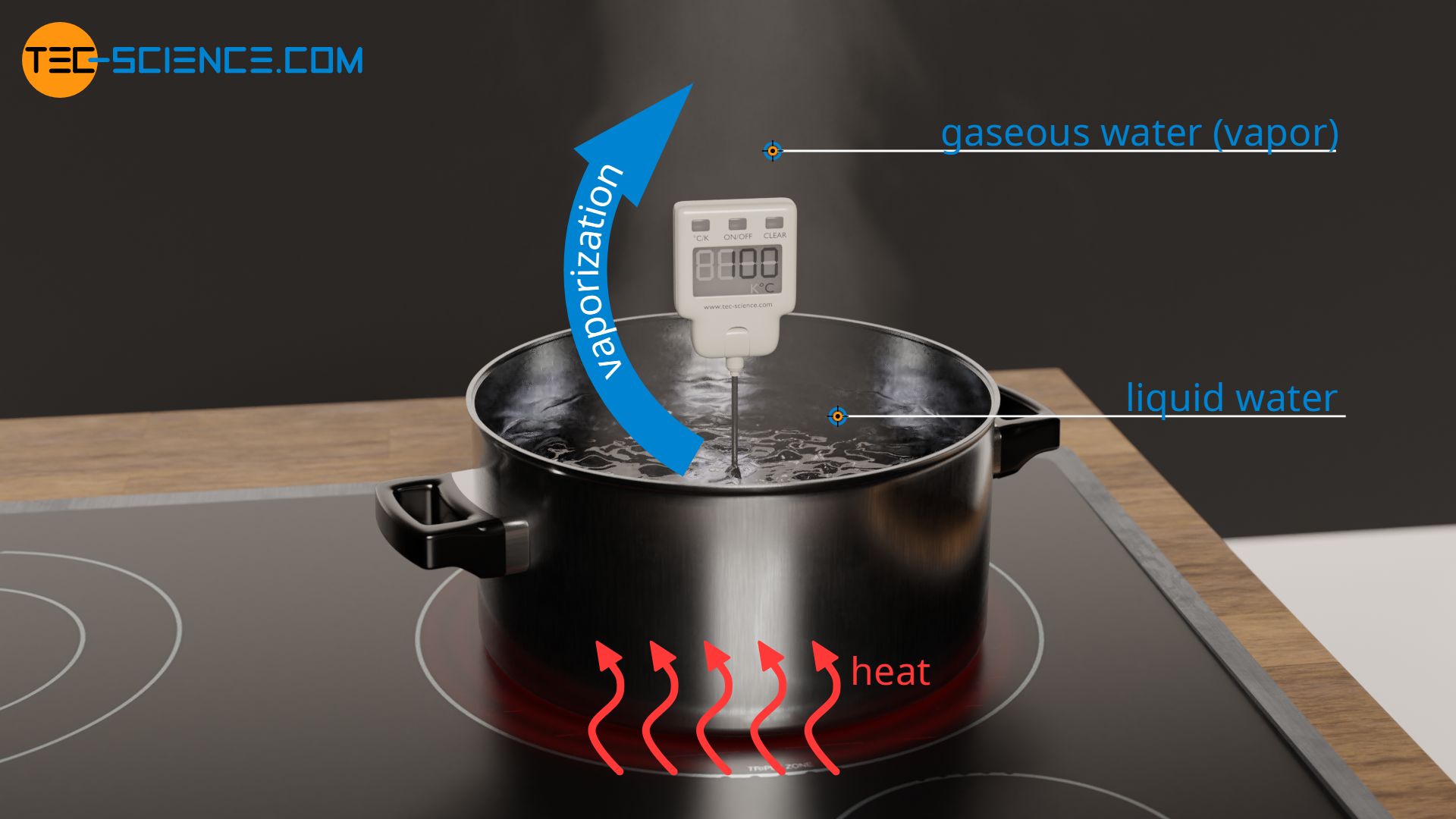
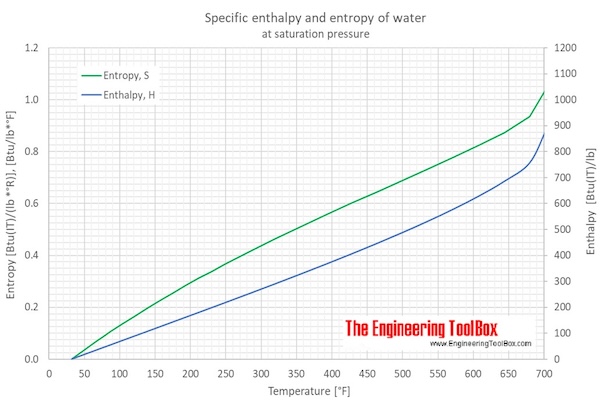
Describe (qualitatively) how standard enthalpy and entropy of vaporization of water will change with temperature. | Homework.Study.com

The enthalpy of vaporization water is 6 186.5 KJ mol-1, the entropy of its vaporization will be- (1) 0.5 KJK-1 mol-1 (2) 1.0 KJK-1 mole-1 (3) 1.5 KJ K-1 mole-1 (4) 2.0 KJK-1 mole-1
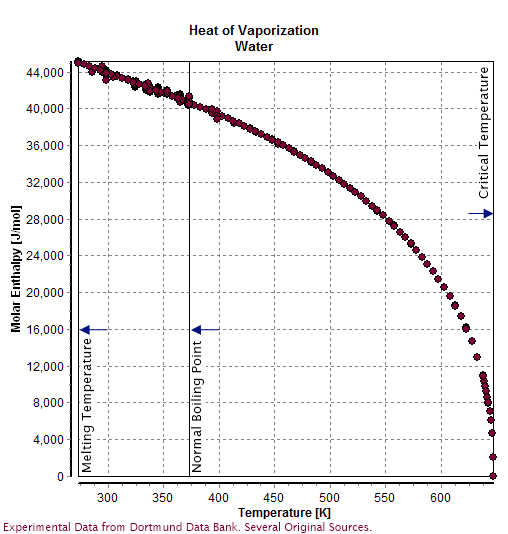
Molar enthalpy of vaporization of water from triple to critical points. | Download Scientific Diagram
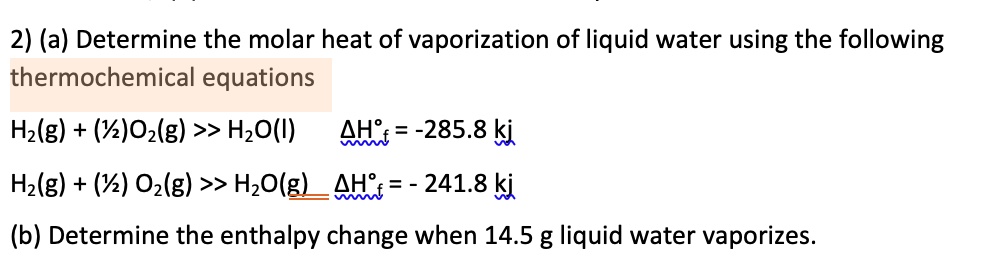
SOLVED: (a) Determine the molar heat of vaporization of liquid water using the following thermochemical equations H2(g) + 1/2O2(g) >> H2O(l) ΔH = -285.8 kJ H2(g) + 1/2O2(g) >> H2O(g) ΔH = -
![standard enthalpy of vapourisation A HO) water 100°C is 40.66 kJmol.The internal energy of vapourisation of water 100°C (in kJmot) is [AIPMT (Prelims)-2012] (2) +40.66 (4) -43.76 funtor is 1 435 kcal/mol. (1) +43.76 (3) +37.56 standard enthalpy of vapourisation A HO) water 100°C is 40.66 kJmol.The internal energy of vapourisation of water 100°C (in kJmot) is [AIPMT (Prelims)-2012] (2) +40.66 (4) -43.76 funtor is 1 435 kcal/mol. (1) +43.76 (3) +37.56](https://toppr-doubts-media.s3.amazonaws.com/images/8783536/2a161f71-6f15-470c-af56-293d004f42a8.jpg)
standard enthalpy of vapourisation A HO) water 100°C is 40.66 kJmol.The internal energy of vapourisation of water 100°C (in kJmot) is [AIPMT (Prelims)-2012] (2) +40.66 (4) -43.76 funtor is 1 435 kcal/mol. (1) +43.76 (3) +37.56

Enthalpy of vaporization of water: (—) Reference fundamental equation... | Download Scientific Diagram

Describe (qualitatively) how standard enthalpy and entropy of vaporization of water will change with temperature. | Homework.Study.com